
Chemistry, 04.07.2019 22:00, jlayne0605
Which substance in this redox reaction is the oxidizing agent? cu + 2agno3 → 2ag + cu(no3)2 a. n b. agno3 c. cu d. no3− e. cu(no3)2

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:30, aedmund1225
Agas has a volume of 0.7 l at 300 mmhg. what would be the new volume at 900 mmhg
Answers: 1


Chemistry, 22.06.2019 12:30, ethanw8973
If 22.5 liters of oxygen reacted with excess of hydrogen, how many liters of water vapor could be produced?
Answers: 3

Chemistry, 23.06.2019 08:00, oopsorry
Determine the number of moles of air present in 1.35 l at 750 torr and 17.0°c. which equation should you use? n=pv/rt what is the number of moles present? ⇒ 0.056 mol a sample of n2 gas occupying 800.0 ml at 20.0°c is chilled on ice to 0.00°c. if the pressure also drops from 1.50 atm to 1.20 atm, what is the final volume of the gas? which equation should you use? v2= p1v1t2/p2t1 what is the final volume of the gas? ⇒ 932 ml these are the answers
Answers: 1
Do you know the correct answer?
Which substance in this redox reaction is the oxidizing agent? cu + 2agno3 → 2ag + cu(no3)2 a. n b...
Questions in other subjects:



Mathematics, 13.01.2022 08:50

Mathematics, 13.01.2022 08:50


Biology, 13.01.2022 08:50



Biology, 13.01.2022 09:00

History, 13.01.2022 09:00


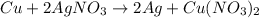
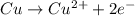
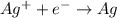
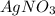 is considered as an oxidizing agent and therefore the correct answer is Option B.
is considered as an oxidizing agent and therefore the correct answer is Option B.



