
Chemistry, 04.07.2019 23:00, BIKRAMlawati9684
What is ph when 4.0 ml of 2.0 m barium hydroxide is added to 10.0 ml of 1.00 m nitric acid? 13.63 0.37 0.85 13.15

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 13:00, carlinryan
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2



Chemistry, 23.06.2019 00:00, PineappleDevil889
2-bromo-2-methylbutane undergoes an e1 elimination reaction in the presence of ethanol. in the next reaction only one of the possible products is represented. although the product shown is not the major product of the reaction, notice that there is more than one way it can be produced. complete the mechanism and draw the missing substances.
Answers: 1
Do you know the correct answer?
What is ph when 4.0 ml of 2.0 m barium hydroxide is added to 10.0 ml of 1.00 m nitric acid? 13.63...
Questions in other subjects:

Mathematics, 01.10.2019 11:30



Mathematics, 01.10.2019 11:30


Mathematics, 01.10.2019 11:30

Social Studies, 01.10.2019 11:30

English, 01.10.2019 11:30

Advanced Placement (AP), 01.10.2019 11:30


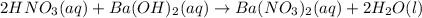
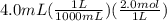
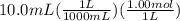
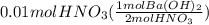
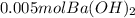
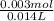
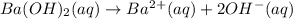
![[OH^-]=2*0.214M](/tpl/images/0051/9161/cdf6f.png) = 0.428M
= 0.428M![pOH=-log[OH^-]](/tpl/images/0051/9161/4fd6b.png)





