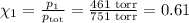Amixture of 60 mol % n-propylcyclohexane and 40 mol % n-propylbenzene is distilled through a simple distillation apparatus; assume that no fractionation occurs during the distillation. the boiling temperature is found to be 157 degrees celsius (760 torr) as the first small amount of distillate is collected. the standard vapor pressures of n-propylcyclohexane and n-propyl-benzene are known to be 769 torr and 725 torr, respectively, at 1567.3 degrees celsius. calculate the percentage of each of the two components in the first few drops of distillate.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:10, goodygoodgirlygirl
Which of the following best describes the formation of plasma?
Answers: 1

Chemistry, 22.06.2019 00:30, TMeansStupidity
Jessica is traveling from miami, florida, to chicago, illinois. using the map, tell one way the land will change during the second half of her trip.
Answers: 1


Chemistry, 22.06.2019 12:00, shifaxoxoxo
What term is applied to a scientist who studies ancient life, including animal and plant fossils a. anthropologist b. dendroclimatologist c. geophysicist d. paleontologist
Answers: 2
Do you know the correct answer?
Amixture of 60 mol % n-propylcyclohexane and 40 mol % n-propylbenzene is distilled through a simple...
Questions in other subjects:






Mathematics, 22.10.2019 23:00


Mathematics, 22.10.2019 23:00

Mathematics, 22.10.2019 23:00


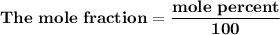
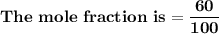
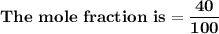
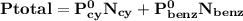
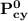 = n-propylcyclohexane vapor pressure
= n-propylcyclohexane vapor pressure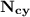 = n-propylcyclohexane mole fraction
= n-propylcyclohexane mole fraction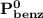 = n-propylbenzene vapor pressure
= n-propylbenzene vapor pressure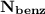 = n-propylbenzene mole fraction
= n-propylbenzene mole fraction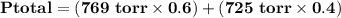
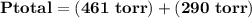

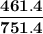
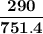
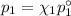 and
and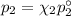
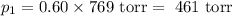
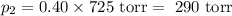
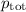 = p₁ + p₂= 461 torr + 290 torr = 751 torr
= p₁ + p₂= 461 torr + 290 torr = 751 torr