The maximum amount of 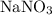 formed from reaction is .
formed from reaction is .
Further Explanation:
The chemical reaction that contains an equal number of atoms of the different elements in the reactant as well as in the product side is known as balanced chemical reaction. The chemical equation is required to be balanced to follow the Law of the conservation of mass.
The steps to balance a chemical reaction are as follows:
Step1: Complete the reaction and write the unbalanced symbol equation.
In the reaction, 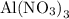 reacts with to form
reacts with to form 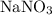 and
and 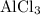 . The unbalanced chemical equation is as follows:
. The unbalanced chemical equation is as follows:
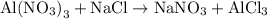
Step2: Then we write the number of atoms of all the different elements that are present in a chemical reaction in the reactant side and product side separately.
On reactant side,
Number of aluminiumatoms is 1.
Number of nitrogen atoms is 3.
Number of oxygen atoms is 6.
Number of sodium atoms is 1.
Number of chlorine atoms is 1.
On product side,
Number of aluminium atoms is 1.
Number of nitrogen atoms is 3.
Number of oxygen atoms is 6.
Number of sodium atoms is 1.
Number of chlorine atoms is 1.
Step3: Initially, we try to balance the number of other atoms of elements except for carbon, oxygen, and hydrogen by multiplying with some number on any side.
To balance the number of atoms we have to multiply 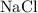 by 3,
by 3, 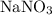 by 3. This gives,
by 3. This gives,
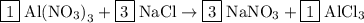
Step 4: After this, we balance the number of atoms of carbon and then hydrogen atom followed by oxygen atoms, but in the reaction no carbon or hydrogen is there and oxygen is already balanced.
Step 5: So the balanced chemical equation is given as follows:
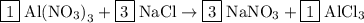
As per stoichiometery, the ratio of moles of 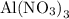 that react with
that react with 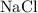 is 1:3. If 1 moles of
is 1:3. If 1 moles of 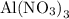 react with 3 moles of
react with 3 moles of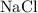 , then 9 moles of
, then 9 moles of 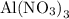 will react with 3 moles of
will react with 3 moles of 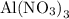 . Hence
. Hence 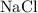 is a limiting reagent.And
is a limiting reagent.And 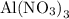 is present in excess.
is present in excess.
So 9 moles of 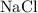 will produce 9 moles of
will produce 9 moles of 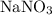 .
.
The formula to calculate the amount of 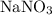 is as follows:
is as follows:
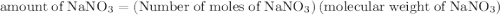
Substitute 9 for number of moles of 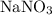 , and
, and 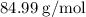 for molecular weight of
for molecular weight of 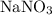 to calculate the amount of
to calculate the amount of 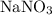 .
.
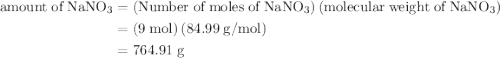
The maximum amount of 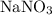 formed from reaction is
formed from reaction is 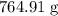 .
.
Learn More:
What are the moles of oxygen formed when 58.6g of
Find the enthalpy of decomposition of 1 mole of MgO:
Answer Details:
Grade: Senior School
Chapter: Chemical Reactions
Subject: Chemistry
Keywords: NaCl, Na(NO3)3, AlCl3, Al(NO3)2, 9 mol, 4 mol, stoiochiometery.


















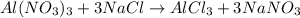
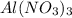 and 9 mol NaCl.
and 9 mol NaCl. 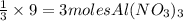
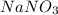 .
.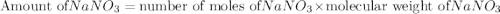
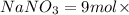 84.99 g/mol = 764.91 grams
84.99 g/mol = 764.91 grams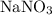 formed from reaction is .
formed from reaction is .
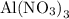 reacts with to form
reacts with to form 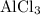 . The unbalanced chemical equation is as follows:
. The unbalanced chemical equation is as follows:
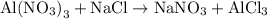
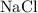 by 3,
by 3, 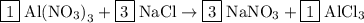
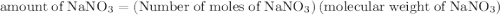
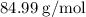 for molecular weight of
for molecular weight of 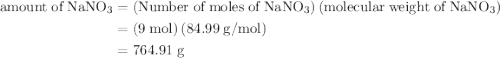
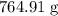 .
.




