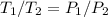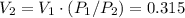Chemistry, 06.07.2019 17:00, miguel454545
Explain how you can use boyle's law to determine the new volume of gas when it's pressure is increased from 170 to 5: 40 the original volume of gas is one leader assume the temperature and number of particles are consistent what is the new volume

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, FloweyFlower
Aballoon inflated with three breaths of air has a volume of 1.7 l. at the same temperature and pressure, what is the volume of the balloon if five more same-sized breaths are added to the balloon? a balloon inflated with three breaths of air has a volume of 1.7 l. at the same temperature and pressure, what is the volume of the balloon if five more same-sized breaths are added to the balloon?
Answers: 3

Chemistry, 22.06.2019 00:30, jamesnaquan132
What is the most stable monatomic ion formed from nitrogen
Answers: 2

Chemistry, 22.06.2019 02:30, rileyeddins1010
List four observations that indicate that a chemical reaction may be taking place
Answers: 1
Do you know the correct answer?
Explain how you can use boyle's law to determine the new volume of gas when it's pressure is increas...
Questions in other subjects:











 Initial Pressure
Initial Pressure  Final Pressure
Final Pressure