Chemistry, 06.07.2019 17:00, allisonchorath6971
How do anion formation and valence electrons relate? atoms gain valence electrons to form anions. b) atoms yield valence electrons to form anions. c) atoms donate valence electrons to form anions. d) atoms relinquish valence electrons to form anions. light is released when an excited electron moves from higher energy level back down to its lower, more stable energy level. the resulting spectrum is a(n) spectrum. a) emission b) absorption c) excitation d) higher energy stars, such as our sun, use fusion to combine hydrogen atoms into helium atoms, and in the process, create energy. as massive stars use the last of their helium fuel, they begin to collapse and temperatures climb high enough to fuse other heavier elements. as elements increase in atomic number, the amount of energy required for fusion to occur also increases. nickel represents the heaviest element that can be produced by fusion due to to the net energy requirements. two atoms of could combine by fusion in order to create nickel. a) hydrogen b) nitrogen c) oxygen d) silicon

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 23.06.2019 03:00, BeeShyanne
Use the half-reactions of the reaction au(oh)3 + hi -> au +i2 +h2o to answer the questions
Answers: 1
Do you know the correct answer?
How do anion formation and valence electrons relate? atoms gain valence electrons to form anions....
Questions in other subjects:

Mathematics, 14.06.2021 15:40







Mathematics, 14.06.2021 15:40

Mathematics, 14.06.2021 15:40


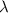 :
: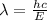 , E = energy, c= speed the light, h= planck constant
, E = energy, c= speed the light, h= planck constant