
What does the pauli exclusion principle state?
a) electrons will be added to an atom at the lowest possible level or sublevel.
b) an orbital can contain two electrons only if all other orbitals at that sublevel are empty.
c) an orbital can contain two electrons only if all other orbitals at that sublevel contain at least one electron.
d) two electrons occupy the same orbital only if they have opposite spins.
e) an orbital can contain two electrons only if all other orbitals at that sublevel are empty.

Answers: 1
Similar questions

Chemistry, 01.07.2019 12:20, ladybugys
Answers: 1

Mathematics, 03.08.2019 03:10, russboys3
Answers: 1

Chemistry, 18.09.2019 17:30, johnross17
Answers: 1

Physics, 13.10.2019 04:30, unknown9263
Answers: 3
Do you know the correct answer?
What does the pauli exclusion principle state?
a) electrons will be added to an atom a...
a) electrons will be added to an atom a...
Questions in other subjects:

History, 24.06.2019 08:00








English, 24.06.2019 08:00

Mathematics, 24.06.2019 08:00


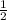 or
or 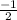 .
.



