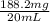Astudent added 32.60 ml of 0.03020 m naoh to a 20.0 ml sample of ginger ale before his sample turned pink. (a) how many moles of naoh did he add to his solution? (b) how many moles of citric acid are in his sample? (c) how many grams of citric acid does this correspond to? (d) how many milligrams? how many mg/ml?

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 15:00, emmalie52
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1


Chemistry, 22.06.2019 22:20, trockout4868
How do cfcs cause ozone depletion? how do cfcs cause ozone depletion? ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule.
Answers: 2
Do you know the correct answer?
Astudent added 32.60 ml of 0.03020 m naoh to a 20.0 ml sample of ginger ale before his sample turned...
Questions in other subjects:








Mathematics, 18.10.2019 18:30

Social Studies, 18.10.2019 18:30


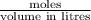
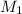 = 0.03020 M
= 0.03020 M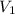 = 32.60 mL= 0.0326 L
= 32.60 mL= 0.0326 L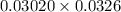 = 0.00098 moles
= 0.00098 moles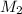 =?
=?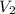 = 20 mL= 0.02 L
= 20 mL= 0.02 L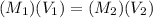 ,
, 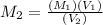
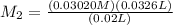 = 0.049 M
= 0.049 M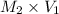 of solution in Liters =
of solution in Liters = 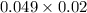 = 0.00098 moles
= 0.00098 moles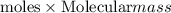
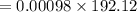 = 0.1882 g
= 0.1882 g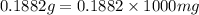 = 188.2 mg (1g = 1000mg)
= 188.2 mg (1g = 1000mg)