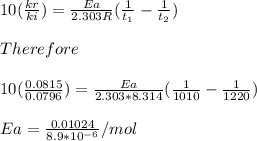Chemistry, 07.07.2019 21:00, madisontrosclair2
Understanding the high-temperature behavior of nitrogen oxides is essential for controlling pollution generated in automobile engines. the decomposition of nitric oxide (no) to n2 and o2 is second order with a rate constant of 0.0796 m−1⋅s−1 at 737∘c and 0.0815 m−1⋅s−1 at 947∘c. you may want to reference (page) section 14.5 while completing this problem. part a calculate the activation energy for the reaction. express the activation energy in kilojoules per mole to three significant digits.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:30, leilanimontes714
Asample of silver (with work function ? = 4.52 ev) is exposed to an ultraviolet light source (? = 200 nm), which results in the ejection of photoelectrons. what changes will be observed if: silver is replaced with copper (? = 5.10 ev) more photoelectrons ejected no photoelectrons are emitted fewer photoelectrons ejected more energetic photoelectrons (on average) less energetic photoelectrons (on average)
Answers: 3

Chemistry, 22.06.2019 17:20, holmesleauja
Which of these features are formed when hot groundwater is forced out through cracks in the earth's surface?
Answers: 2

Chemistry, 23.06.2019 09:10, amandapill
In a 28 g serving of cheese curls there are 247mg of sodium. how much sodium is in a 12.5 ounce bag
Answers: 1

Chemistry, 23.06.2019 15:30, tacosloco4629
Light travels through space at 186,282 miles per second and it takes about 1.3 seconds for light to travel from the moon to earth. which of the following is the correct method of finding the distance, in miles, between the moon and earth? add 186,282 and 1.3 divide 186,282 by 1.3 multiply 186,282 by 1.3 subtract 1.3 from 186,282
Answers: 1
Do you know the correct answer?
Understanding the high-temperature behavior of nitrogen oxides is essential for controlling pollutio...
Questions in other subjects:

Chemistry, 03.08.2019 12:00




Biology, 03.08.2019 12:00

History, 03.08.2019 12:00

Social Studies, 03.08.2019 12:00

Mathematics, 03.08.2019 12:00

Mathematics, 03.08.2019 12:00

Mathematics, 03.08.2019 12:00