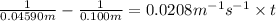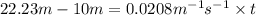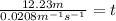Chemistry, 08.07.2019 15:30, jaymaxron512
For the reaction, a → b, the rate constant is 0.0208 m-1 sec-1. how long would it take for [a] to decrease from 0.100 to 0.0450 m?

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 19:30, xxaurorabluexx
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2 (g) + 2h2o (g) → 4hcl (g) + o2 (g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound concentration at equilibrium cl2 0.55m h2o 0.57m hcl 0.53m o2 0.34m calculate the value of the equilibrium constant kc for this reaction. round your answer to 2 significant digits.
Answers: 2

Chemistry, 22.06.2019 21:00, Janznznz4012
Once similarity and one difference between a mixture of elements and a mixture of compounds
Answers: 3

Chemistry, 22.06.2019 21:20, 50057543
Phosgene (carbonyl chloride), cocl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: co(g) cl2(g)⇌cocl2(g) carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 477 ∘c . at equilibrium, the concentrations were measured and the following results obtained: gas partial pressure (atm) co 0.830 cl2 1.30 cocl2 0.220 what is the equilibrium constant, kp, of this reaction
Answers: 2
Do you know the correct answer?
For the reaction, a → b, the rate constant is 0.0208 m-1 sec-1. how long would it take for [a] to de...
Questions in other subjects:





English, 18.09.2019 19:00




History, 18.09.2019 19:00


![\frac{dx}{dt}=k\times[A]^{n}](/tpl/images/0066/0583/78eab.png)
![\frac{1}{[A]_{t}}-\frac{1}{[A]_{o}} = kt](/tpl/images/0066/0583/3b1d5.png) (second order formula)
(second order formula)