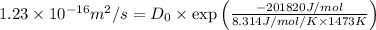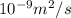Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:00, macylen3900
Ineed this asap part i: scientific method what is the difference between science and pseudoscience? what is the scientific method?
Answers: 2

Do you know the correct answer?
The diffusivity of ni in mgo is 1.23 x 10-16 m2/s at 1200˚c and 1.45 x 10-14 m2/s at 1800˚c. calcula...
Questions in other subjects:











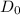 = 1.7658 ×
= 1.7658 × 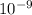
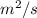
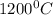 ,
, 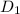 = 1.23 ×
= 1.23 × 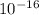
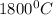 ,
, 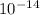
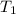 =
= 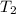 =
= 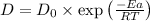 ..........(1)
..........(1)![ln\frac{D_{1}}{D_{2}}=\frac{Ea}{R}\left [\frac{1}{T_{2}}-\frac{1}{T_{1}} \right ]](/tpl/images/0068/0569/c61c4.png) ........(2)
........(2)![ln\frac{1.23\times 10^{-16}}{1.45\times 10^{-14}}=\frac{Ea}{8.314}\left [\frac{1}{2073}-\frac{1}{1473} \right ]](/tpl/images/0068/0569/685a3.png)