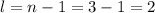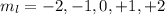The four sets of quantum numbers for nickel (n, l, 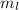 ,
, 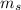 ) are (3, 2, 0, -1/2).
) are (3, 2, 0, -1/2).
To find the quantum numbers of Ni, we need to look at its electron configuration. The atomic number of Ni is 28, so its electronic configuration is:
1s² 2s² 2p⁶ 3s² 3p⁶ 3d⁸ 4s² → [Ar] 3d⁸ 4s²
The last electron is in the d-shell, so let's find the quantum numbers.
Principal quantum number, n
Since the shell of the last electron is 3 (3d⁸), this number 3 corresponds to the quantum number n (n=3).
Azimuthal quatum number, l
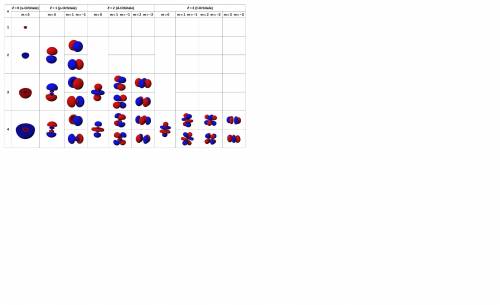
This quantum number is related to the shape of the orbital and is given by n-1. The types of shapes for the values of l are:
l = 0 corresponds to the orbital's shape "s"
l = 1 corresponds to the orbital's shape "p"
l = 2 corresponds to the orbital's shape "d"
l = 3 corresponds to the orbital's shape "f "
The shell of the last electron of Ni is 3 (3d⁸), so l is:
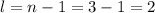
Magnetic quantum number,
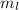
This quantum number varies from -l to +l. For Ni, l = 2, so the possible values of 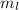 are:
are:
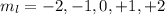
This means that we have 5 orbitals, which can have a maximum of 2 electrons (Pauli exclusion principle). Electron filling in those orbitals goes in order from -2 to +2, so for Ni, the last electron is in the 0 orbital.
Hence, the value of 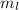 is 0.
is 0.
Spin quantum number,
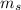
This number can have two possible values: +1/2 (spin-up) or -1/2 (spin-down). For the filling of the electrons in the orbitals, we start for the positive values (+1/2, spin-up) and then we culminate with the negative ones (-1/2, spin-down), to complete the filling of two electrons per orbital. Hence, for Ni, the value of 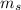 is -1/2 (spin-down).
is -1/2 (spin-down).
Therefore, the four sets of quantum numbers (n, l, 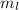 ,
, 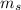 ) for nickel are (3, 2, 0, -1/2).
) for nickel are (3, 2, 0, -1/2).
You can learn more about quantum numbers here: link
I hope it helps you!















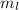 ,
, 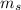 ) are (3, 2, 0, -1/2).
) are (3, 2, 0, -1/2).