Chemistry, 10.07.2019 05:30, cynthiagutierrez65
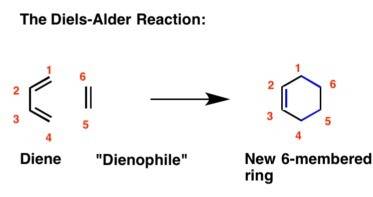
The internal reaction temperature (in other words, the temperature of the reaction mixture inside the reaction flask) of this diels-alder experiment may be specified by the term reflux. a. what was the internal reaction temperature, in °c? b. how was the internal reaction temperature achieved and maintained?

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 18:00, kyllow5644
Answer asap need to be answered by wednesday morning explain how a buffer works, using an ethanoic acid / sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 3

Chemistry, 22.06.2019 20:00, ahnorthcutt4965
Acm ruler with main graduations from 1 to 10 from left to right there are 10 secondary graduations between each of the main graduations there is a line that begins. at the left end of the ruler 10 secondary graduations to the left of the “1 main graduation the right end of the line ends on the eighth secondary graduation to the right of 3 how long is the line
Answers: 1
Do you know the correct answer?
The internal reaction temperature (in other words, the temperature of the reaction mixture inside th...
Questions in other subjects:


Mathematics, 05.11.2019 05:31

Mathematics, 05.11.2019 05:31


History, 05.11.2019 05:31


Mathematics, 05.11.2019 05:31


History, 05.11.2019 05:31

Mathematics, 05.11.2019 05:31


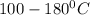 . At this temperature, the bonds are broken and rearranged.
. At this temperature, the bonds are broken and rearranged.