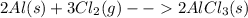Chemistry, 10.07.2019 05:30, Ciarrathereal
Solid aluminum metal and diatomic chlorine gas react spontaneously to form a solid product. give the balanced chemical equation (including phases) that describes this reaction.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, megaaan214p61pb7
Which compounds have the empirical formula ch2o? a. c2h4o2 b. c3h6o3 c. ch2o2 d. c5h10o5 e. c6h12o6
Answers: 3

Chemistry, 22.06.2019 06:30, AleciaCassidy
Identify the missing numbers below to show the result of multiplying the numbers (1.6 × 10-19)(5.0 × 106) = c × 10d
Answers: 1


Chemistry, 22.06.2019 14:00, hammackkatelyn60
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3
Do you know the correct answer?
Solid aluminum metal and diatomic chlorine gas react spontaneously to form a solid product. give the...
Questions in other subjects:



History, 12.11.2020 22:10

Mathematics, 12.11.2020 22:10

Mathematics, 12.11.2020 22:10




Mathematics, 12.11.2020 22:10

Mathematics, 12.11.2020 22:10


 .
.