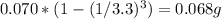In this experiment, 0.070 g of caffeine is dissolved in 4.0 ml of water. the caffeine is then extracted from the aqueous solution three times, each time using 2.0 ml fresh methylene chloride. calculate the total amount of caffeine that can be extracted into the three portions of methylene chloride (caffeine has a distribution coefficient of 4.6 between methylene chloride and water).

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, salvadorperez26
Suppose that during that icy hot lab 65,000 j of energy were transferred to 450 g of water at 20°c what would have have been the final temperature of the water
Answers: 2



Chemistry, 22.06.2019 17:00, emma3216
In a heat engine of 1000 j of heat enters the system and the piston does 500 j of work what is the final internal energy of the system if the inital energy was 2000 j we have to do all of these down here 1)write the equation 2)list out your know variables 3)plug the numbers into the equations 4)solve 5)write your solution statemtn that includes inital energuy and final energuy added
Answers: 1
Do you know the correct answer?
In this experiment, 0.070 g of caffeine is dissolved in 4.0 ml of water. the caffeine is then extrac...
Questions in other subjects:

Physics, 05.05.2020 11:55

Mathematics, 05.05.2020 11:55






Biology, 05.05.2020 11:55



 .
.