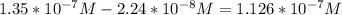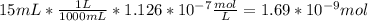Chemistry, 10.07.2019 21:00, deanlmartin
In a typical analysis, 15 ml of an aqueous solution containing an unknown amount of acetylcholine had a ph of 7.65. when incubated with acetylcholinesterase, the ph of the solution decreased to 6.87. assuming there was no buffer in the assay mixture, determine the number of moles of acetylcholine in the 15 ml sample.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:00, annarain2004
Asample of gas occupies 17 ml at –112°c. what volume does the sample occupy at 70°c a. 10.6 ml b. 27 ml c. 36 ml d. 8.0 ml you
Answers: 1

Chemistry, 22.06.2019 04:00, tifftifftiff5069
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1

Chemistry, 22.06.2019 04:30, aleilyg2005
There is a single path for electrons. the current decreases when additional resistors are added. the current will be the same in each resistor. these statements best describe a(n) circuit.
Answers: 3

Chemistry, 22.06.2019 17:30, latezwardjr15
In a heat of an engine, if 700 j enters the system, and the piston does 400 j of work what is the final internal (thermal) energy of the system if the initial energy is 1,500 j
Answers: 2
Do you know the correct answer?
In a typical analysis, 15 ml of an aqueous solution containing an unknown amount of acetylcholine ha...
Questions in other subjects:


Mathematics, 22.02.2021 19:40

Mathematics, 22.02.2021 19:40

Social Studies, 22.02.2021 19:40

Spanish, 22.02.2021 19:40






![[H_{3}O^{+}]=10^{-7.65}=2.24*10^{-8} M](/tpl/images/0074/6124/2f1c4.png)
![[H_{3}O^{+}]=10^{-6.87}=1.35*10^{-7} M](/tpl/images/0074/6124/010e5.png)