
The solubility of oxygen gas in water at 40 ∘c is 1.0 mmol/l of solution. what is this concentration in units of mole fraction? the solubility of oxygen gas in water at 40 is 1.0 of solution. what is this concentration in units of mole fraction? 1.80×10−5 1.80×10−2 1.00×10−2 5.55×10−2 1.00×10−6

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:30, luhmimi17
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 13:30, yasiroarafat12
How many moles is 14.5 cm^3 of platinum? the density of platinum is 21.45 g/cm^3.
Answers: 1

Chemistry, 22.06.2019 23:00, edgar504xx
What is the solubility-product constant of barium sulfate, baso4, if a saturated solution is 1.03 ´ 10-5 m?
Answers: 3
Do you know the correct answer?
The solubility of oxygen gas in water at 40 ∘c is 1.0 mmol/l of solution. what is this concentration...
Questions in other subjects:











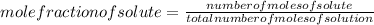 -(1)
-(1)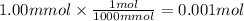
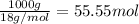
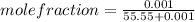
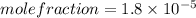
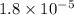 .
.




