
Chemistry, 11.07.2019 07:00, hannahliebl2000
What is the molarity of a salt solution made by dissolving 560 mg of nacl in 4 ml of water? what is the osmolarity of this solution?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, orlando19882000
Ahypothrticalax type of ceramic material is known to have a density of 2.10 g/cm3 and a unit cell of cubic symmetry with a cell edge length of 0.57 nm. the atomic weights of the a and x elements are 28.5and 30.0 g/mol, respectively. on the basis of this information, which of the following crystal structures is (are) possible for this material: sodium chloride, cesium chloride, or zinc blende
Answers: 1

Chemistry, 22.06.2019 12:00, Unknowndragon42
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 19:00, innocentman69
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1
Do you know the correct answer?
What is the molarity of a salt solution made by dissolving 560 mg of nacl in 4 ml of water? what is...
Questions in other subjects:



Mathematics, 10.02.2021 02:00


Mathematics, 10.02.2021 02:00


English, 10.02.2021 02:00


Mathematics, 10.02.2021 02:00

Mathematics, 10.02.2021 02:00


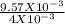 = 2.393 M.
= 2.393 M.



