
Bromine reacts with nitric oxide to form nitrosyl bromide as shown in this reaction: br2(g) + 2 no(g) → 2 nobr(g) a possible mechanism for this overall reaction is shown below. no(g) + br2(g) br2(g) (fast step; keq = k1/k−1) k2 nobr(g) + no(g) → 2 nobr(g) (slow step) what is the rate law for formation of nobr in terms of reactants based on this mechanism

Answers: 1
Similar questions


Chemistry, 01.07.2019 00:00, DwayneLeonard618
Answers: 1

Do you know the correct answer?
Bromine reacts with nitric oxide to form nitrosyl bromide as shown in this reaction: br2(g) + 2 no(...
Questions in other subjects:








Computers and Technology, 03.10.2021 02:30


Mathematics, 03.10.2021 02:30


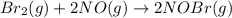
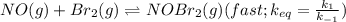
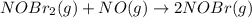 (slow step
(slow step 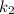 )
)![r_{1}=k_{1}[NO][Br_{2}]-k_{-1}[NOBr_{2}]](/tpl/images/0077/5023/9bc26.png)
![r_{2}=k_{2}[NOBr_{2}] [NO]](/tpl/images/0077/5023/6e659.png)
![[NOBr_{2}]](/tpl/images/0077/5023/48931.png) takes place in this reaction.
takes place in this reaction.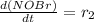
![k_{2}[NOBr_{2}][NO]](/tpl/images/0077/5023/433e8.png) (1)
(1)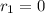
![k_{1}[NO][Br_{2}]= k_{-1}[NOBr_{2}]](/tpl/images/0077/5023/8345a.png)
![[NOBr_{2}] = \frac{k_{1}}{k_{-1}}[NO][Br_{2}]](/tpl/images/0077/5023/ee42f.png)
![\frac{d(NOBr)}{dt}=k_{2}[NOBr_{2}][NO]](/tpl/images/0077/5023/ff99b.png)
![k_{2} \frac{k_{1}}{k_{-1}}[NO][Br_{2}][NO]](/tpl/images/0077/5023/f5111.png)
![\frac{k_{1}k_{2}}{k_{-1}}[NO]^{2}[Br_{2}]](/tpl/images/0077/5023/427a8.png)
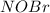 in terms of reactants is given by
in terms of reactants is given by 



