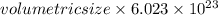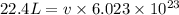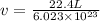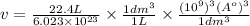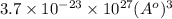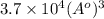Calculate the volumetric size of a water molecule in water vapor at normal conditions, assuming 1 mole of the vapor occupies 22.4 l, as if the vapor were an ideal gas. give answer in angstroms, two significant digits. do not write down units in your answer. calculate the volumetric size of a water molecule in water vapor at normal conditions, assuming 1 mole of the vapor occupies 22.4 l, as if the vapor were an ideal gas. give answer in angstroms, two significant digits. do not write down units in your answer.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:00, 2019reynolds
Which sentence about particles in matter is true? a. atoms are present in solids and liquids but not in gases. b. the particles of matter are in constant motion. c. the same kinds of atoms are found in different elements. d. when a solid changes to a liquid, the sizes of the particles change.
Answers: 1

Chemistry, 22.06.2019 12:30, emmalybrown
A50.0 ml sample of gas at 20.0 atm of pressure is compressed to 40.0 atm of pressure at constant temperature. what is the new volume? 0.0100 ml 0.325 ml 25.0 ml 100. ml
Answers: 1

Chemistry, 22.06.2019 15:30, elizabethprasad2
The reactions of photosynthesis occur in the of plant cell? a. mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1

Do you know the correct answer?
Calculate the volumetric size of a water molecule in water vapor at normal conditions, assuming 1 mo...
Questions in other subjects:





English, 19.02.2020 10:05





English, 19.02.2020 10:10


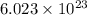 molecules of water.
molecules of water.