
Chemistry, 11.07.2019 19:00, hayesvolcano
A1.42-g sample of a pure compound with formula m2so4 was dissolved in water and treated with an ex- cess of aqueous calcium chloride, resulting in the precipi- tation of all the sulfate ions as calcium sulfate. the pre- cipitate was collected, dried, and found to weigh 1.36 g. determine the atomic mass of m and identify m.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:00, alevans7144
Why do sodium and neon have vastly different chemical and physical properties despite having similar atomic masses?
Answers: 2

Chemistry, 21.06.2019 17:10, codeyhatch142
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1

Do you know the correct answer?
A1.42-g sample of a pure compound with formula m2so4 was dissolved in water and treated with an ex-...
Questions in other subjects:


Social Studies, 29.07.2021 05:00


Mathematics, 29.07.2021 05:00


Chemistry, 29.07.2021 05:00

History, 29.07.2021 05:00




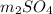 with calcium chloride can be shown as-
with calcium chloride can be shown as-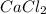 →
→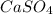 ↓+2mCl. The molecular weight of
↓+2mCl. The molecular weight of 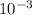 moles of calcium sulfate.
moles of calcium sulfate.



