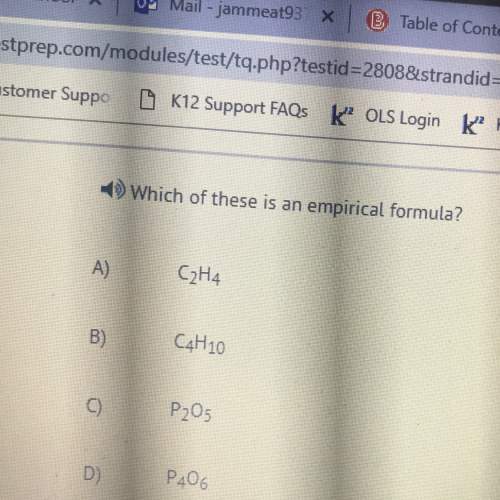
Chemistry, 12.07.2019 09:30, qudoniselmore0
Complete combustion of 1.5 g of fructose a sugar that contains carbon, hydrogen, and oxygen yields 2.2 g of carbon dioxide and .9 g of water. determine the empirical formula of fructose

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:30, acaciacoats
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2

Chemistry, 22.06.2019 17:30, shookiegriffin
I'm learning about the periodic tables and what each subject's configuration is. for example, hydrogen is 1s^1, but i don't understand how you get that. can someone me understand how to figure out how to figure this out? sorry if the question makes no sense, but it would really a lot if you could me understand! you so much if you can!
Answers: 1

Do you know the correct answer?
Complete combustion of 1.5 g of fructose a sugar that contains carbon, hydrogen, and oxygen yields 2...
Questions in other subjects:

Mathematics, 27.10.2020 02:30

Mathematics, 27.10.2020 02:30



Social Studies, 27.10.2020 02:30

Biology, 27.10.2020 02:30

Mathematics, 27.10.2020 02:30

Engineering, 27.10.2020 02:30


Mathematics, 27.10.2020 02:30


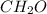 .
.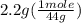 = 0.05 mole
= 0.05 mole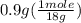 = 0.05 mole
= 0.05 mole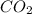 has one C means the mole ratio of
has one C means the mole ratio of 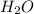 has two H means the mole ratio of
has two H means the mole ratio of 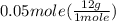 = 0.6 g
= 0.6 g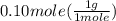 = 0.1g
= 0.1g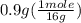 = 0.056 moles
= 0.056 moles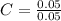 = 1
= 1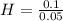 = 2
= 2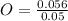 = 1
= 1