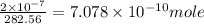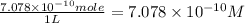Answers: 1
Similar questions


Do you know the correct answer?
The concentration of the alkane c20h42 (fm 282.56) in a particular sample of rainwater is 0.2 ppb. a...
Questions in other subjects:









Social Studies, 01.04.2021 17:20


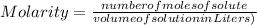 -(1)
-(1)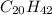 is determined as:
is determined as: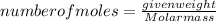 -(2)
-(2)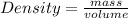
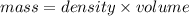 - (3)
- (3)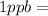
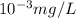
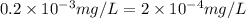
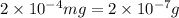 (As 1 mg =
(As 1 mg = 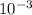 g).
g).