
Chemistry, 12.07.2019 13:00, daebreonnakelly
Calculate the ph for the following weak acid. a solution of hcooh has 0.14m hcooh at equilibrium. the ka for hcooh is 1.8×10−4. what is the ph of this solution at equilibrium? express the ph numerically.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:40, loveoneonly9153
Consider this initial-rate data at a certain temperature for the reaction described by
Answers: 1

Chemistry, 22.06.2019 14:00, ashlynneboogs0056
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3

Chemistry, 22.06.2019 18:30, bibiansolis
The table lists the lattice energies of some compounds. compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf. the lattice energy increases as the cations get larger, as shown by lif and licl. the lattice energy decreases as cations get smaller, as shown by nacl and naf. the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3

Chemistry, 22.06.2019 19:50, mikaylaaaaa
If a gas has an initial pressure of 101kpa and a volume of 10l, then it expands to a volume of 20l, what is the new pressure?
Answers: 2
Do you know the correct answer?
Calculate the ph for the following weak acid. a solution of hcooh has 0.14m hcooh at equilibrium. th...
Questions in other subjects:




English, 14.02.2022 06:00




Mathematics, 14.02.2022 06:10

Mathematics, 14.02.2022 06:10


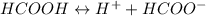
![Ka=\frac{[H^+][HCOO^-]}{[HCOOH]}](/tpl/images/0081/0231/b2152.png)
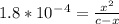
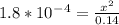
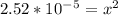
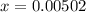
![[H^+]](/tpl/images/0081/0231/07acb.png) .
. ![pH=-log[H^+]](/tpl/images/0081/0231/15713.png)




