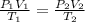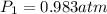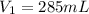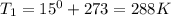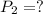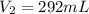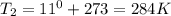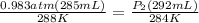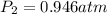Chemistry, 14.07.2019 02:30, ethanyayger
Astudent collects 285 ml of o2 gas at a temperature of 15°c and a pressure of 0.983 atm. the next day, the same sample occupies 292 ml at a temperature of 11°c. what is the new pressure of the gas? 0.946 atm 1.00 atm 0.704 atm

Answers: 2
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 22:00, Porciabeauty6788
The diagrams to the right show the distribution and arrangement of gas particles in two different containers. according to kinetic-molecular theory, which of the following statements is true? check all that apply. if the temperatures of both containers are equal, container a has greater pressure than container b. if the volume of container a decreased, its pressure would decrease. if the pressure in both containers is equal, container a has a lower temperature than container b.
Answers: 2

Chemistry, 22.06.2019 22:30, angelagonzalesownus1
Which statement best summarizes the importance of ernest rutherford’s gold foil experiment? it proved that all of john dalton’s postulates were true. it verified j. j. thomson’s work on the atomic structure. it showed that an electron circles a nucleus in a fixed-energy orbit. it showed that a nucleus occupies a small part of the whole atom.
Answers: 1
Do you know the correct answer?
Astudent collects 285 ml of o2 gas at a temperature of 15°c and a pressure of 0.983 atm. the next da...
Questions in other subjects:



Mathematics, 25.06.2019 10:00