
Chemistry, 14.07.2019 04:30, spooderfaxy7813
The acid-dissociation constant at 25.0 °c for hypochlorous acid (hclo) is 3.0 ⋅ 10−8. at equilibrium, the molarity of h3o+ in a 0.055 m solution of hclo is

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, terrancebest
Which is a chemical property of iron? a. it forms iron oxide (rust) when exposed to moisture and air. b. it is a gray–black metal that is hard to the touch. c. it has a melting point of 2795°f (1536°c). d. it is a good conductor of heat
Answers: 2

Chemistry, 22.06.2019 12:40, valenzueladomipay09u
How does concentration affect reaction rate
Answers: 2

Chemistry, 22.06.2019 17:00, jazmine8194
Reduction is a reaction which results in a in electrons and a in positive charge of the atom or ion 1) a- loss 1) b- gain 2) a-increase 2) b-decrease
Answers: 1

Chemistry, 22.06.2019 22:00, cooljariel11
Give more examples of this type of heat transfer:
Answers: 1
Do you know the correct answer?
The acid-dissociation constant at 25.0 °c for hypochlorous acid (hclo) is 3.0 ⋅ 10−8. at equilibrium...
Questions in other subjects:


Mathematics, 11.11.2020 18:10

History, 11.11.2020 18:10

History, 11.11.2020 18:10

Mathematics, 11.11.2020 18:10


Mathematics, 11.11.2020 18:10




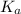 value for the dissociation of HOCl is
value for the dissociation of HOCl is 






