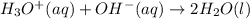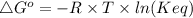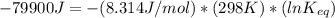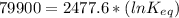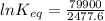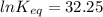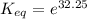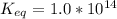Chemistry, 14.07.2019 10:30, samantha636
The standard free energy change for the formation of two moles of h2o(l) in a strong acid–strong base neutralization reaction at 25°c is -79.9kj. calculate the equilibrium constant for the reaction. see equation 11.1. h3o+(aq) + oh-(aq) = 2 h2o (l)

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:30, tbeck225
If a solution is considered basic, then a) the hydroxide ion and hydronium ion concentrations are equal. b) the hydroxide ion concentration is less than the hydronium ion concentration. c) the hydronium ion concentration is greater than the hydroxide ion concentration. d) the hydroxide ion concentration is greater than the hydronium ion concentration.
Answers: 1

Chemistry, 22.06.2019 07:50, mckinleesmomp6qj1e
Which of the following electromagnetic waves can create ions?
Answers: 2

Chemistry, 22.06.2019 16:50, lilblackbird4
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3
Do you know the correct answer?
The standard free energy change for the formation of two moles of h2o(l) in a strong acid–strong bas...
Questions in other subjects:

Biology, 01.10.2019 04:30

Mathematics, 01.10.2019 04:30

Mathematics, 01.10.2019 04:30


Mathematics, 01.10.2019 04:30


Mathematics, 01.10.2019 04:30

History, 01.10.2019 04:30

Chemistry, 01.10.2019 04:30