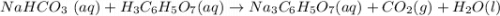Chemistry, 14.07.2019 13:00, amandaparrish2323
Alka-seltzer is an antacid that is dissolved in water before use. for an experiment, three tablets of alka-seltzer are dissolved in three beakers, each containing 50 ml of water: beaker a's water is 10° c; beaker b's water is 50° c; and beaker c's water is 80° c. which answer correctly orders the reaction rates of the three beakers from fastest to slowest reaction? beaker a > beaker b > beaker c beaker c > beaker b > beaker a beaker b > beaker a > beaker c beaker a = beaker b = beaker c

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, kichensides
Asample of aluminum foil contains 8.60 × 1023 atoms. what is the mass of the foil?
Answers: 1

Chemistry, 22.06.2019 19:10, aamu15
Which statement correctly describes the phosphate ion, ? it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge on the phosphorus atom. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge on the phosphorus atom.
Answers: 3

Chemistry, 23.06.2019 01:30, sheldonwaid4278
Magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. trial 1: trial 2: data mass of empty crucible with lid trial 1: 26.688 trial 2: 26.681 mass of mg metal, crucible, and lid trial 1: 26.994 trial: 2 26.985 mass of mgo, crucible, and lid trial 1: 27.188 trial 2: 27.180
Answers: 1

Chemistry, 23.06.2019 07:30, jessicawelch25
In a laboratory determination of the atomic weight of tin, a sample of tin is weighed in a crucible. nitric acid is added, and the reaction proceeds to give a hydrated tin(iv)oxide plus no2and h2o. the hydrated tin(iv)oxide is then heated strongly and reacts as follows: sno2.xh2o(s)sno2(s)+ xh2o(g)the sno2is finally cooled and weighed in the crucible. explain the effect on the calculated atomic weight of tin that would result from each of the following experimental errors: (a)considerable spattering occurs when the nitric acid is added to the tin.(b)the hydrated tin(iv)oxide is not heated sufficiently to change it completely to tin oxide.
Answers: 2
Do you know the correct answer?
Alka-seltzer is an antacid that is dissolved in water before use. for an experiment, three tablets o...
Questions in other subjects:

Mathematics, 10.11.2020 21:00



History, 10.11.2020 21:00

Physics, 10.11.2020 21:00

Mathematics, 10.11.2020 21:00


Biology, 10.11.2020 21:00

Health, 10.11.2020 21:00

Mathematics, 10.11.2020 21:00