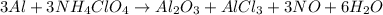Chemistry, 14.07.2019 15:00, franky2871
The reusable booster rockets of the u. s. space shuttle employ a mixture of aluminum and ammonium perchlorate for fuel. a possible equation for this reaction is3al1s213nh4clo41s2hal2o31s21alcl3 1s213no1g216h2o1g2what mass of nh4clo4 should be used in the fuel mixture for every kilogram of al?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:30, noathequeen
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 13:30, xojade
Which statements are true concerning mineral formation? check all that apply. the slower the cooling, the larger the crystals. the faster the cooling, the smaller the crystals. crystals formed from magma are smaller than crystals formed from lava. minerals can only form in solutions when the solution is heated deep underground. when a solution cools, elements and compounds leave the solution and crystallize as minerals. minerals formed from hot water solutions can form narrow channels in the surrounding rock.
Answers: 1

Do you know the correct answer?
The reusable booster rockets of the u. s. space shuttle employ a mixture of aluminum and ammonium pe...
Questions in other subjects:

Mathematics, 03.05.2021 20:40



Mathematics, 03.05.2021 20:40


Computers and Technology, 03.05.2021 20:40



Spanish, 03.05.2021 20:40