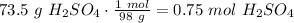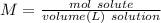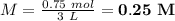Chemistry, 14.07.2019 21:00, tiffxnnyyy
A3.0 l solution contains 73.5 g of h2so4. calculate the molar concentration of the solution.0.25 m0.44 m0.75 m4.0 m

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:00, lorenaandreahjimenez
The answer for #3 is c but i don't know why
Answers: 1

Chemistry, 22.06.2019 04:30, akeemedwards12
Both josef loschmidt and amedeo avogadro contributed to our understanding of basic molecular numbers, sizes, and reaction ratios. neither scientist discovered “avogadro’s number” in the form we use it today (6.02 x 10 23). still, there’s a controversy over the name. research the contributions from these two scientists and read about how avogadro’s number got its name. briefly state what you think this number should be called, providing key details of each scientist’s contributions to this concept and a solid rationale for your case in naming the number.
Answers: 2

Chemistry, 22.06.2019 07:00, haydjanggg6578
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2
Do you know the correct answer?
A3.0 l solution contains 73.5 g of h2so4. calculate the molar concentration of the solution.0.25 m0....
Questions in other subjects:


Chemistry, 01.12.2020 17:40

Mathematics, 01.12.2020 17:40







Mathematics, 01.12.2020 17:40


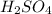 is:
is: