
Chemistry, 13.10.2019 09:30, hannahcolleen4895
Fill in the blanks:
1. during a chemical reaction, the sum of the of the reactants and products remain unchanged.
2. in a pure chemical compound, elements are always present in a proportion by mass.
3. clusters of atoms that act as an ion are called ions.
4. in ionic compounds, the charge on each ion is used to determine the the compound.
5. the avogadro constant is defined as the number of atoms in exactly of carbon-12
6. mass of 1 mole of a substance is called its
7. the abbreviation used for length names of elements are termed as their
8. a chemical formula is also known as a
9. those ions which are formed from single atoms are called
10. ionic compounds are formed by the combination between an
11. the valency of an ion is to the charge on the ion.
12. mole is a link between the and
13. the si unit of amount of a substance is
true / false:
1. formula mass of na2o is 62 amu.
2. those particles which have more or less electrons than the normal atoms are called ions
.
3. formula for sulphur dioxide is so3.
4. molar mass of ethyne (c2h2) is 26 g/mol.
5. 22 gm of co2 consists of 1 mole.
6. number of molecules in 32 gram of oxygen is 6.02 x 1023.
7. water is an atom.
8. formula for sulphur dioxide is so2.
9. clusters of atoms that act as an ion is called lopyatomic ion.
10. mass of 1 mole of a substance is called its formula mass.
11. in a pure chemical compound, elements are always present in a definite proportion by mass.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, darg3990rgp2t0r2
Does anyone know a lot about how to: - calculate mass of magnesium metal - calculate the actual yield of magnesium oxide - calculate the theoretical yield of mgo - calculate the percent yield of mgo - determine the percent yield of mgo - determine the average percent yield of mgo i had to do an online lab and its asking these questions but i have no idea where to start or how to be able to find these things. i can post the chart of the data from the lab or if you can tell me exactly how i can find each.
Answers: 3


Chemistry, 22.06.2019 11:30, claudr03
If we compare and contrast electromagnetic waves with sound waves, all but one statement is true. that is a) sound waves require a medium to travel while electromagnetic waves do not. b) electromagnetic waves can travel through the vacuum of space while sound waves cannot. c) electromagnetic waves must have a medium in which to travel, but sound waves can travel anywhere. eliminate d) sound waves must bounce off of matter in order to travel while electromagnetic waves do not require matter to be present.
Answers: 3
Do you know the correct answer?
Fill in the blanks:
1. during a chemical reaction, the sum of the of the reactants and...
1. during a chemical reaction, the sum of the of the reactants and...
Questions in other subjects:

Mathematics, 23.08.2021 17:40

Mathematics, 23.08.2021 17:40

Mathematics, 23.08.2021 17:40

Mathematics, 23.08.2021 17:40

Mathematics, 23.08.2021 17:40






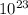 is defined as the number of atoms in exactly 12 g of carbon-12
is defined as the number of atoms in exactly 12 g of carbon-12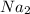 O is 62 amu True
O is 62 amu True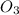 . False
. False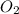 as the name di suggests two molecules.
as the name di suggests two molecules.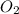 contains 1 C atom and 2 O atoms (12) + (16 X 2) = 44
contains 1 C atom and 2 O atoms (12) + (16 X 2) = 44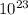 . False
. False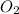 true.
true.




