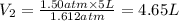Chemistry, 16.07.2019 10:30, honeysenpai6
The pressure of 5.0 l of gas increases from 1.50 atm to 1240 mm hg. what is the final volume of the gas, assuming constant temperature?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:10, akatsionis25
When will le chatelier's principle come into effect? at the beginning of a reaction, when there are only reactants when a reaction has reached chemical equilibrium when a catalyst is added to a reaction mixture when a reaction is occurring but not yet at equilibrium
Answers: 3

Chemistry, 21.06.2019 22:50, Catracho3619
Blank allows you to do calculations for situations in which only the amount of gas is constant a)boyle's law b)combined gas law c)ideal gas law d)dalton's law
Answers: 1


Chemistry, 22.06.2019 21:30, kawaiiblurainbow
Which of the following changes will decrease the total amount of gaseous solute able to be dissolved in a liter of liquid water? (2 points) decreasing temperature decreasing pressure decreasing surface area decreasing solute concentration
Answers: 1
Do you know the correct answer?
The pressure of 5.0 l of gas increases from 1.50 atm to 1240 mm hg. what is the final volume of the...
Questions in other subjects:


Mathematics, 29.05.2020 21:03

Biology, 29.05.2020 21:03


Social Studies, 29.05.2020 21:03






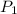 = 1.50 atm
= 1.50 atm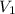 = 5.0 L
= 5.0 L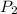 = 1240 mmHg = 1.612 atm
= 1240 mmHg = 1.612 atm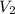
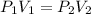 (Constant temperature)
(Constant temperature)