
Chemistry, 18.07.2019 07:00, nekobaby75
The poh of a solution is 10.75. what is the concentration of ions in the solution? use mc013-1.jpg. 3.162 mc013-2.jpg m 1.778 mc013-3.jpg m 1.075 mc013-4.jpg 102 m 5.623 mc013-5.jpg 1010 m

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 16:30, ccispoppin12
Asample of freon gas has a volume of 2.23 liters, a pressure of 4.85 kpa, and a temperature of -1.36°c. calculate the volume at a pressure of 1.38 kpa and a temperature of 5.5°c. (show work)
Answers: 1

Chemistry, 22.06.2019 21:20, 50057543
Phosgene (carbonyl chloride), cocl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: co(g) cl2(g)⇌cocl2(g) carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 477 ∘c . at equilibrium, the concentrations were measured and the following results obtained: gas partial pressure (atm) co 0.830 cl2 1.30 cocl2 0.220 what is the equilibrium constant, kp, of this reaction
Answers: 2
Do you know the correct answer?
The poh of a solution is 10.75. what is the concentration of ions in the solution? use mc013-1.jpg...
Questions in other subjects:


English, 05.10.2020 01:01


Mathematics, 05.10.2020 01:01

English, 05.10.2020 01:01




Mathematics, 05.10.2020 01:01


![[OH^-] =10^{-10.75}=1.778\times 10^{-11}M](/tpl/images/0103/1172/da2fe.png)
![[OH^-]](/tpl/images/0103/1172/b2910.png) ions , lesser is the pOH and vice versa.
ions , lesser is the pOH and vice versa.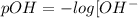
![10.75=-log[OH^-]](/tpl/images/0103/1172/69f4a.png)
![[OH^-] =10^{(-10.75)}=1.778\times 10^{-11}M](/tpl/images/0103/1172/b9026.png)

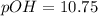
![pH=-\log [H^+]](/tpl/images/0103/1172/37e81.png) .........(1)
.........(1)![pOH=-\log [OH^-]](/tpl/images/0103/1172/1fac1.png) ........(2)
........(2)![10.75=-\log [OH^-]](/tpl/images/0103/1172/c006c.png)
![[OH^-]=10^{-10.75}](/tpl/images/0103/1172/2175a.png)
![[OH^-]=1.778\times 10^{-11}M](/tpl/images/0103/1172/05884.png)





