
Chemistry, 21.11.2019 21:31, nathanstern21
Pl
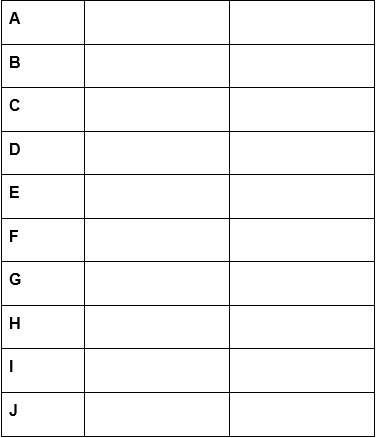
5. the following equations represent part of a radioactive decay series:
1. 92238u -> a + 24he
2. a -> 91234pa + b
3. 91234pa -> c + –10e
4. c -> 90230th + 24he
5. 90230th -> d + 24he
6. d -> 86222rn + e
7. 86222rn -> 84218po + f
8. 84218po -> g + 24he
9. g -> 83214bi + h
10. 83214bi -> i + –10e
11. i -> j + 24he
fill in the table with the correct particle or element, and describe the types of radiation given off in the series.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 16:30, ddmoorehouseov75lc
Correct relationship between molecular formula and empirical formula
Answers: 1

Chemistry, 22.06.2019 18:00, ameliaxbowen7
Heat is the total potential energy of a substance that can be transferred. true false
Answers: 1

Chemistry, 22.06.2019 18:30, losalobos46
The number of moles of a given mass of a substance can be found without knowing its molecular formula or molar mass. true false
Answers: 1
Do you know the correct answer?
Pl
5. the following equations represent part of a radioactive decay series:
1. 92238u...
5. the following equations represent part of a radioactive decay series:
1. 92238u...
Questions in other subjects:








Chemistry, 01.05.2021 18:10

History, 01.05.2021 18:10






