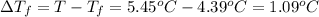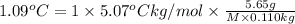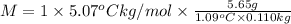Chemistry, 20.07.2019 15:30, emmanuelmashao5504
Asolution is made by dissolving 5.65 g of an unknown molecular compound in 110.0 g of benzene froze at 4.39 oc. what is the molar mass of the solute if pure benzene has a freezing point of 5.45 oc and the kf value of benzene is 5.07 oc/m

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, arodavoarodavo
The table lists pressure and volume values for a particular gas. which is the best estimate for the value of v at p = 7.0 × 103 pascals?
Answers: 3

Chemistry, 22.06.2019 06:30, coreyslotte
Use examples from the article to explain one positive and one negative effect that chemistry has had on society.
Answers: 2

Do you know the correct answer?
Asolution is made by dissolving 5.65 g of an unknown molecular compound in 110.0 g of benzene froze...
Questions in other subjects:

Physics, 16.09.2019 18:20


History, 16.09.2019 18:20


Mathematics, 16.09.2019 18:20

Biology, 16.09.2019 18:20

Biology, 16.09.2019 18:20


History, 16.09.2019 18:20


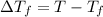
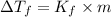
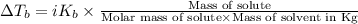
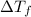 =Depression in freezing point
=Depression in freezing point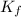 = freezing point constant of solvent= 5.07°C/m
= freezing point constant of solvent= 5.07°C/m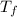 =4.39 °C
=4.39 °C