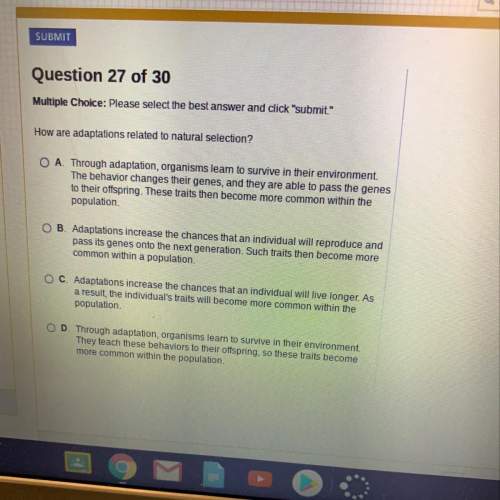
Biology, 13.11.2019 23:31, maddieberridgeowud2s
If δg = δh - tδs, when is the overall free energy change (δg) in a reaction most likely to be negative (meaning that the reaction is exergonic)?
a. when products have lower potential energy and higher entropy than reactants
b. when products have higher potential energy and lower entropy than reactants
c. when products have lower potential energy and lower entropy than reactants
d. when products have higher potential energy and higher entropy than reactants

Answers: 3
Other questions on the subject: Biology

Biology, 21.06.2019 23:30, chonawilson4
What is the importantence of the carbon and nitretan cycles in the environment
Answers: 1

Biology, 22.06.2019 06:40, keenansimpkinsoy0oqc
Which is not a type of symmetry? a. asymmetryb. radial symmetryc. bilateral symmetryd. lateral symmetry
Answers: 3
Do you know the correct answer?
If δg = δh - tδs, when is the overall free energy change (δg) in a reaction most likely to be negati...
Questions in other subjects:


History, 29.07.2019 16:30


History, 29.07.2019 16:30

Business, 29.07.2019 16:30



Mathematics, 29.07.2019 16:30

History, 29.07.2019 16:30